FEMALE REPRODUCTIVE SYSTEM
- Jump to LINKS:
- OVARY
- OVARIAN FOLLICULAR DEVELOPMENT
- CORPUS LUTEUM
- UTERUS
- UTERINE (FALLOPIAN) TUBES
- VAGINA
- PLACENTA
- MAMMARY GLANDS
OVERVIEW
The female reproductive system consists of 2 ovaries, 2 oviducts (also known as uterine or fallopian tubes), the uterus, vagina, external genitalia and 2 mammary glands. The system undergoes cyclic changes in structure and function between menarche, when the first menses appear, and the menopausal period, when the reproductive system undergoes involution.
Image: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, p. 464
1-THE OVARY
The ovary is a rounded body approx. 3 x 1.5 cm long and 1 cm thick. It is encapsulated by the tunica albuginea, a dense layer of connective tissue which is covered by the germinal epithelium (Ovarian surface epithelium), a layer of simple cuboidal epithelium. The ovarian follicles, which enclose the oocytes, are primarily embedded in the cortical region of the ovary while the medullary portion contains a rich vascular bed. The ovarian stroma consists of characteristic spindle shaped fibroblasts that respond to hormonal stimuli, reticular fibers and ground substance.
1.1 – OOGENESIS
Oogenesis is a specialized process, resulting in the production of one mature ovum and three nuclear masses, the polar bodies. During oogenesis, unequal division of the cytoplasm occurs, so that the fertilized egg will have adequate organelles and nourishment to sustain during its journey down the fallopian tube.
During embryonic development, about 1700 primordial germ cells migrate to the ovary where they undergo mitosis and give rise to approx. 7 million oogonia. The oogonia enlarge, enter the prophase of the first meiotic division, and are now called primary oocytes. (Review meiosis). More than 99% of the primary oocytes undergo atresia (degeneration). Approx. 2 million primary oocytes survive to birth; 300,000 survive to puberty; and 400-500 are ovulated, usually one per month, during the reproductive lifetime of a woman.
2 – OVARIAN FOLLICULAR DEVELOPMENT
Follicular maturation involves changes in the oocyte, the follicular cells and the surrounding stroma. Four types of follicles can be distinguished in the postpubertal ovarian cortex: Primordial follicles, primary follicles, secondary (antral) follicles; and mature (Graafian) follicles.
Image: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, p. 465
2.1 – TYPES OF OVARIAN FOLLICLES
2.1A. PRIMORDIAL FOLLICLES
Primordial follicles are the principle follicles present before birth and are also present in the adult ovary. Each consists of a primary oocyte enveloped by a single layer of flattened follicular cells.

Image: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, p. 465
2.1B. PRIMARY FOLLICLES
The primary follicle is characterized by growth of the oocyte, an enveloping layer of cuboidal follicular cells, and the zona pellucida, an acellular glycoprotein layer between the oocyte and the follicular cells. The follicular cells extend cytoplasmic processes through the zona and form gap junctions with the oocytes. Nutrients and regulatory factors that control oocyte maturation are transmitted through these junctions. The oocyte, in turn, secretes factors that influence the subsequent proliferation and differentiation of the follicle cells. The stroma surrounding the follicle becomes organized into the theca folliculi. Development of the primary follicle involves proliferation of the follicular cells into a multilayered zona granulosa and differentiation of the theca folliculi into the theca interna and theca externa.


Image: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, p. 465
2.1C. SECONDARY FOLLICLES
The secondary follicle exhibits further enlargement of the zona granulosa and a follicular antrum in which follicular fluid accumulates. The cumulus oophorus is the mass of granulosa cells surrounding the oocyte. The theca externa forms a capsule-like stroma surrounding the follicle, while the theca interna will form a functional cell layer necessary for estrogen synthesis. The theca interna differentiates into typical steroid secreting cells and produces an estrogen precursor (androstenedione) which is converted into estrogen by the granulosa cells containing the aromatase enzyme. (Below, a visual summary of estrogen synthesis by the theca interna and granulosa cells is provided).

Image: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, p. 465
Two-Cell Model of Estrogen Synthesis in Follicular Phase of Menstrual Cycle:
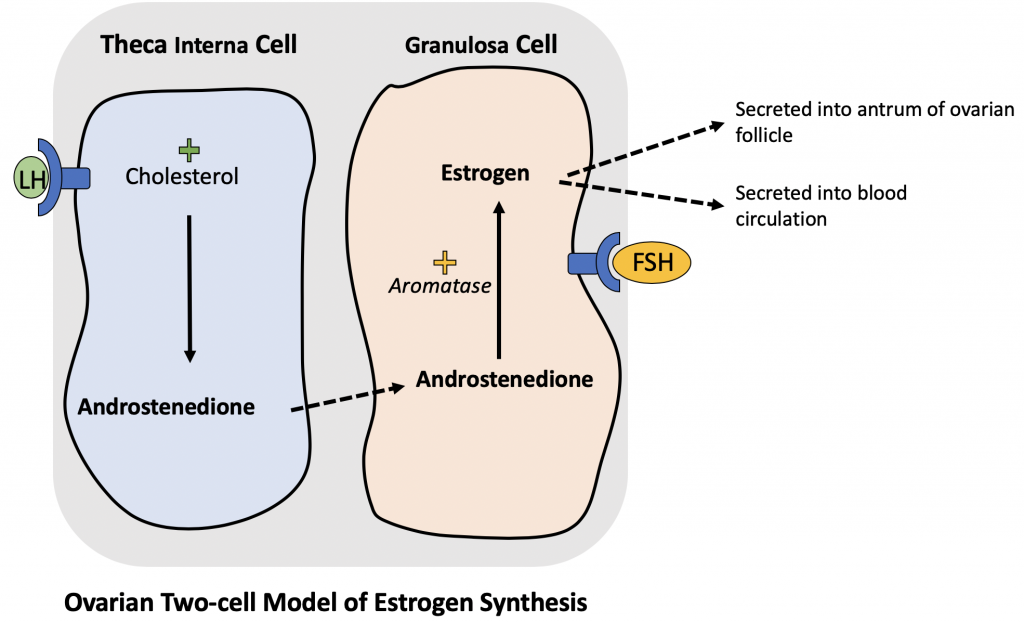
Histophysiology of Estrogen Synthesis:
Prior to ovulation, the uterus must undergo proliferation in order to re-establish a functional endometrium for potential implantation of a fertilized oocyte. During the follicular phase, estrogen is synthesized through a two-cell system formed by the the theca interna and granulosa cells. Under stimulation from LH, the theca interna cell converts cholesterol to androstenedione. Androstenedione then diffuses into the granulosa cell. Under stimulation from FSH, the granulosa cell primes the enzyme aromatase to convert the androstenedione into estrogen. Estrogen is secreted into the blood as well as accumulates within the follicular antrum of the developing follicle. The estrogen produced by the follicles stimulates the uterus to proliferate the endometrium in preparation for implantation of the fertilized oocyte.
2.1D. MATURE (GRAAFIAN) FOLLICLE
The mature (Graafian) follicle is characterized by an enlarged follicular antrum and thinned granulosa layer. The diminished cumulus oophoros surrounding the oocyte is called the corona radiata. It is during this stage that the primary oocyte completes the first meiotic division, extrudes the first polar body, and forms the second metaphase spindle. It is now in the secondary oocyte stage.
Ovulation involves rupture of the follicle and release of the secondary oocyte, with the corona radiata. Fertilization provides the stimulus to complete the second meiotic division.

Image: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, p. 465
2.1E. ATRETIC FOLLICLE
Primordial follicles undergo atresia starting in the fetal stages and this process continues till menopause. Beginning at puberty approximately 20 primordial follicles begin to develop at each cycle but usually only a single follicle completes maturation. The remaining follicles undergo atresia which can occur at any stage of follicular development.
2.2 CORPUS LUTEUM
Following ovulation the follicle collapses and fills with a blood clot (corpus hemorragicum). The cells of the granulosa layer and theca interna form a temporary endocrine gland called the corpus luteum. Since the corpus luteum is a gland, it must become heavily vascularized in order for the hormones to enter the systemic circulation to reach their targets (i.e. endometrium of the uterus). The cells of the theca interna, now called theca lutein cells, continue to synthesize estrogen precursors (androstenedione). The granulosa cells, now called granulosa lutein cells continue to receive and convert androstenedione to estrogen using the aromatase enzyme. Importantly, the granulosa lutein cells, now heavily vascularized, can receive cholesterol from the blood and convert it into another hormone called progesterone. As long as the corpus luteum remains functional, it can continually secrete progesterone to maintain the endometrial lining of the uterus until implantation occurs. If implantation of a fertilized egg fails to occur after ovulation, the corpus luteum will degrade and form the corpus albicans. After ovulation, the corpus luteum will only remain functional for approximately 14 days.

Image: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, p. 465
i) CORPUS LUTEUM –> CORPUS ALBICANS
If implantation of a fertilized egg on the uterine endometrium does not occur (pregnancy), the corpus luteum will undergoes autolysis, and is replaced by a fibrous tissue mass called the corpus albicans. This corpus luteum remains viable for approximately 10-14 days after ovulation.
ii) CORPUS LUTEUM (OF PREGNANCY)
When pregnancy occurs the corpus luteum continues to secrete high levels of estrogen and progesterone. This secretion is promoted by a hormone, human chorionic gonadotropin, which is secreted by the developing placenta. The corpus luteum is the major steroid producing organ during the first months of pregnancy and plays a major role in maintaining the fetus within the uterus. Once the placenta is fully established at about three months, the role of the corpus luteum as an endocrine organ becomes less significant. The corpus luteum of pregnancy resembles the corpus luteum of the ovarian cycle in structure but is much larger.
3 – ENDOCRINE FUNCTION
Production of the female sex hormones is regulated by the gonadotrophic hormones, FSH (follicle stimulating hormone), and LH (luteinizing hormone). The gonadotrophins (LH, FSH) are synthesized in the anterior pituitary and their release is controlled by Gonadotropin Releasing Hormone (GNRH), which is liberated by the hypothalamus. FSH stimulates the growth of ovarian follicles and synthesis of estrogens by the theca lutein and granulosa cells. LH promotes ovulation and the formation of the corpus luteum through differentiation of the granulosa cells which secrete progesterone.
Both estrogen and progesterone are involved in negative feedback loops to the hypothalamus and anterior pituitary. Estrogen inhibits FSH secretion; progesterone inhibits LH secretion. Estrogen also operates in a positive feedback loop to stimulate release of GNRH, thereby indirectly stimulating an increased production of progesterone.
The relationship of the ovarian cycle to hormonal activity is as follows: The follicular phase, occupying the first 14 days of the cycle, is characterized by rapidly increasing estrogen levels produced by the developing follicle. The mid-cycle is characterized by a surge of gonadotrophins which stimulates ovulation. Finally, the luteal phase is marked by continued estrogen and increasing progesterone production by the corpus luteum.
4 – UTERINE (FALLOPIAN) TUBES
The uterine tube is a musculomembranous tube with a mucosa that is arranged in longitudinal folds that resemble a labyrinth in cross section. The inner lining consists of a simple columnar epithelial layer composed of secretory peg cells and ciliated cells. The thick mucus film which covers the epithelium is moved in the direction of the uterus by ciliary action and the peristaltic movements of the muscle layer. These movements propel the embryo towards the uterus. The lamina propria is composed of loose connective tissue and the muscle layer consists of criss-crossing bundles of smooth muscle separated by loose connective tissue.
5 – UTERUS
Most of the outer wall of the uterus is composed of an perimetrium (Serosa), which is the portion of the broad ligament covering the superior aspect of the uterus. The middle layer, the myometrium, consists of 4 poorly defined layers of smooth muscle. The endometrium, or uterine mucosa, consists of epithelium, connective tissue stroma, and blood vessels. Importantly, this layer undergoes dramatic cyclic changes during the reproductive years in preparation for implantation of a fertilized egg. (See below for important details regarding the specific layers and function of the endometrium)
5.1 – ENDOMETRIUM
The endometrium is divided into an uppermost stratum functionalis which is sloughed off at menstruation and replaced during each menstrual cycle. The underlying stratum basalis is retained during menstruation and provides the cells that regenerate the endometrium. The stratum basalis is nourished by straight arteries that form an extensive plexus whereas the stratum functionalis is supplied by coiled (spiral) arteries that extend towards the surface with almost no branching.

Image: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, p. 477
5.2 – MENSTRUAL CYCLE
Estrogen and progesterone cause the endometrium to undergo structural changes during the menstrual cycle. These changes are designed to provide an easily penetrable highly vascular tissue with an abundant supply of glycogen to facilitate implantation and nourish the embryo.
By convention the beginning of the menstrual cycle is taken to begin with the menstrual phase (first 4 days of the cycle). During the subsequent proliferative phase (days 5-14; estrogenic) the cells in the glands in the stratum basalis undergo proliferation and reconstitute the glands and epithelial lining of the endometrium while at the same time cellular proliferation in the connective tissue reconstitutes the lamina propria. At the end of the proliferative phase, straight glands predominate in the endometrial mucosa and their cells are accumulating glycogen. This growth phase of the endometrium is controlled by estrogen synthesized by the developing ovarian follicles.
The secretory phase (days 15-28; progesteronal) starts after ovulation and is controlled by progesterone (as well as estrogen) produced by the corpus luteum. This phase is characterized by the development of tortuous uterine glands that produce glycogen-rich secretions & tortuous spiral arteries.
If implantation fails to occur the increasing levels of progesterone act via a negative feedback mechanism to suppress LH release by the anterior pituitary. Suppression of LH causes involution of the corpus luteum and, since the endometrium cannot be maintained in the absence of progesterone, it is sloughed off. The spiral arteries play an important role in this process. Reduced progesterone levels causes them to undergo constriction and the resulting ischemia causes progressive disintegration of the endometrium.

Image: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, p. 479
6 – VAGINA
The vagina consists of a mucosa, muscular layer and fibrous adventitial layer. The epithelium of the mucosa is stratified squamous; the lamina propria is rich in elastic fibers and lacking glands; and the muscular layer is composed of longitudinal and circular smooth muscle fibers. The fibrous adventitia, which is also rich in elastic fibers, connects the vagina with adjacent structures. The high content of elastic tissue and muscle allows expansion of the vaginal wall during parturition.
The vaginal epithelium undergoes cyclic changes during the menstrual cycle. Under the stimulus of estrogen the epithelial cells synthesize glycogen which is converted into lactic acid by bacteria in the vagina. The resultant acid environment probably plays an important role in maintaining a suitable bacterial flora.
7 – PLACENTA
The placenta has a maternal component (Decidua Basalis) and a fetal component (Chorionic Plate with Chorionic Villi). The placenta establishes a route of exchange for gases (oxygen, carbon dioxide), urea (fetus), nutrients, and antibodies between the mother and the developing fetus.
Maternal Component: consists of the decidua basalis, which consists of a modified portion of the endometrium, in which, polyhedral cells (Decidua Cells) are found. These cells contain large amounts of glycogen and lipids to support of the developing fetus.
Fetal Component: consists of chorionic villi, which are fetal finger-like processes that invade the decidua basalis and rupture the spiral arteries located within. After rupture of the spiral arteries, the blood vessels empty their contents into the intervillous space, the empty spaces found between the choronic villi.
7.1 – Cells of the Chorionic Villus:
Syncytiotrophoblasts: the outer layer of cells with indistinct cell borders. The syncytiotrophoblasts invade the endometrium and secrete Human Chorionic Gonatropin hormone (HCG) that stimulates the corpus luteum to continue its production of progesterone to maintain the endometrial-lining during pregnancy.
Cytotrophoblasts: an inner layer cells found adjacent to the syncytiotrophoblasts. These cells consist of a single layer of cells with distinct cell borders.
Mesenchymal connective tissue cells: thin, spindle-like cells that stain poorly.
Endothelial Cells of Fetal Blood Vessels: typical endothelial cells line fetal blood vessels and capillaries within the chorionic villus. Fetal blood vessels consist of several nucleated red blood cells, which are red blood cell precursors of the normoblast (AKA. erythroblast) series.
7.1 – Blood- Placental Barrier
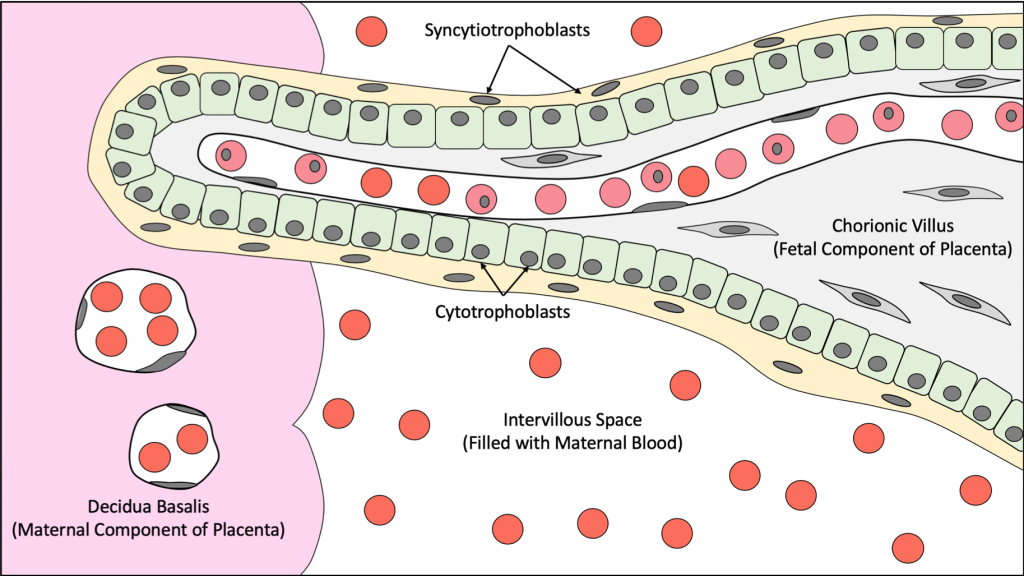
The structures that form the blood-placental barrier are:
- Syncytiotrophoblasts
- Cytotrophoblasts
- Mesenchymal connective tissue cells
- Endothelial cells of fetal blood vessels
8 – MAMMARY GLANDS
Mammary glands secrete milk which consist of proteins, lipids, lactose, antibodies and other nutrients, like vitamin K,to provide proper nourishment for the newborn baby.
The mammary glands consist of compound tubuloalveolar glands found in approximately 15-20 lobes radiating from the nipple. These lobes are separated from each other by adipose and connective tissues. Each lobe is drained by its own lactiferous duct leading directly to the nipple. A dilated region, the lactiferous sinus, stores milk before opening onto the surface of the nipple. The non-secreting mammary glands of the inactive breast has the same basic architectural features of the active secreting mammary glands during pregnancy.
The lactiferous ducts near opening to the skin are lined by keratinized stratified squamous epithelium. The lactiferous sinus and duct are lined by stratified cuboidal epithelium and become simple cuboidal epithelium as the ducts become smaller in size.
8.1 – Lactating (Active) Mammary Glands
Surging levels of progesterone and estrogen during pregnancy stimulate the in-active underdeveloped glands and their associated duct system to grow. As pregnancy progresses, the glandular tissue proliferates and secretes a protein-rich fluid called, Colostrum. Colostrum is rich in proteins and antibodies (IgA) for nutrition and passive immunity of the newborn. Colostrum is secreted during pregnancy and up to 3 days after child birth.
Prolactin from the acidophils of the anterior pituitary gland stimulate the glandular tissue to stop secreting colostrum and begin secreting breast milk, which is a primary feature of the lactating breast.
The alveoli are composed of a simple cuboidal epithelium surrounded by myoepithelial cells, which contract to expel milk, and a basement membrane. Alveolar cells secrete lipids and proteins from their apical surfaces. Milk lipids are exocytosed from the apical cell surface as “blebs”, which contain lipid droplets surrounded by a thin layer of cytoplasm (Apocrine secretion). Milk proteins are secreted directly into the lumen without taking a portion of the cytoplasm with it (Merocrine secretion).

Image: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, p. 486
Images: Gartner & Hiatt (2007). Color Textbook of Histology: Saunders Elsevier, pp 463-488

